Query String: HYDROXYPROGESTERONE CAPROATE
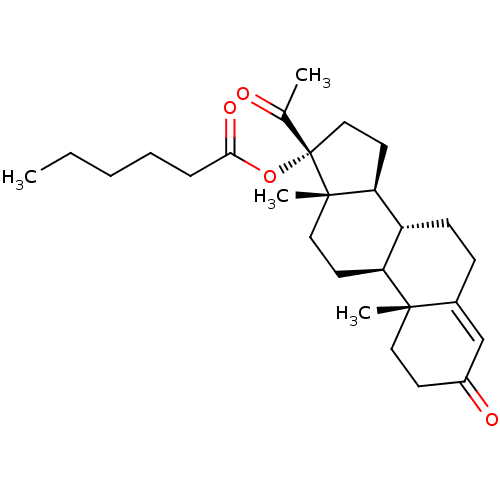 MLS000028438 cid_169870 BDBM70293 SMR000058336 17-alpha-hydroxy-progesterone caproate HYDROXYPROGESTERONE CAPROATE
MLS000028438 cid_169870 BDBM70293 SMR000058336 17-alpha-hydroxy-progesterone caproate HYDROXYPROGESTERONE CAPROATE Rutin caproate BDBM237188
Rutin caproate BDBM237188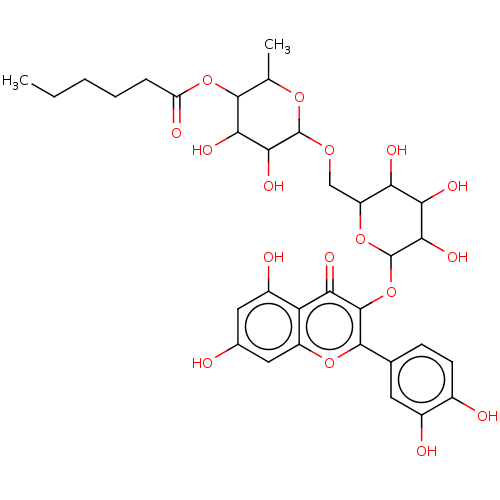 BDBM237186 Rutin caproate Rutin capronate
BDBM237186 Rutin caproate Rutin capronate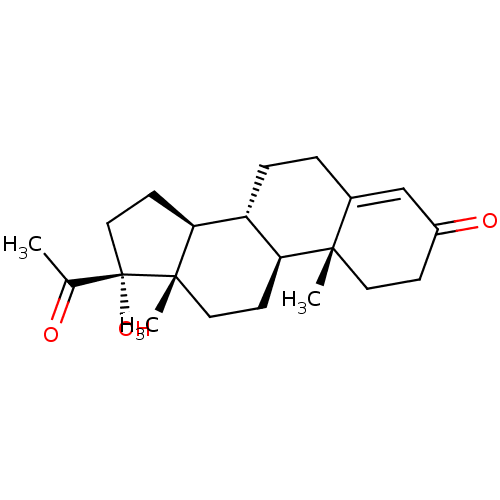 BDBM50423511 HYDROXYPROGESTERONE Delalutin
BDBM50423511 HYDROXYPROGESTERONE Delalutin BDBM50442531 11BETA-HYDROXYPROGESTERONE
BDBM50442531 11BETA-HYDROXYPROGESTERONE n-caproate n-hexylate BDBM50269982 CH3-[CH2]4-COO(-) pentanecarboxylate 1-hexanoate capronate caproate nPnCO2 anion pentylformate hexanoic acid hexoate 1-pentacarboxylate n-hexoate hexylate 1-pentanecarboxylate butylacetate n-Hexanoate hexanoate
n-caproate n-hexylate BDBM50269982 CH3-[CH2]4-COO(-) pentanecarboxylate 1-hexanoate capronate caproate nPnCO2 anion pentylformate hexanoic acid hexoate 1-pentacarboxylate n-hexoate hexylate 1-pentanecarboxylate butylacetate n-Hexanoate hexanoate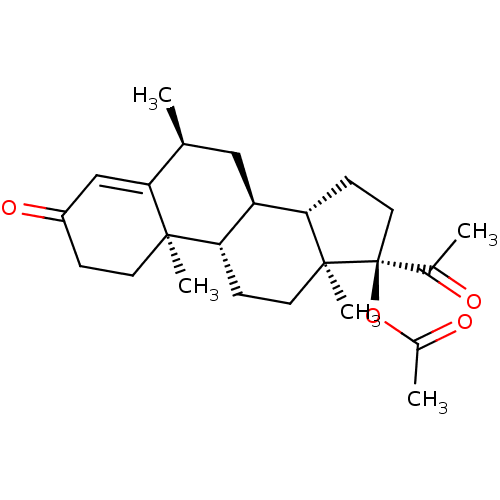 Methylacetoxyprogesterone US9271961, MPA MEDROXYPROGESTERONE 6-alpha-Methyl-17-alpha-hydroxyprogesterone acetate Medroxyprogesterone 17-acetate (6alpha)-6-methyl-3,20-dioxopregn-4-en-17-yl acetate Medroxyacetate progesterone 6alpha-Methyl-17alpha-hydroxyprogesterone acetate medroxyprogesterone acetate 17-Acetoxy-6alpha-methylprogesterone 17alpha-Hydroxy-6alpha-methylprogesterone acetate 6-alpha-Methyl-17-alpha-acetoxyprogesterone 6alpha-Methyl-17-acetoxy progesterone Metigestrona BDBM50067678 (6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-dione 6alpha-Methyl-4-pregnene-3,20-dion-17alpha-ol acetate
Methylacetoxyprogesterone US9271961, MPA MEDROXYPROGESTERONE 6-alpha-Methyl-17-alpha-hydroxyprogesterone acetate Medroxyprogesterone 17-acetate (6alpha)-6-methyl-3,20-dioxopregn-4-en-17-yl acetate Medroxyacetate progesterone 6alpha-Methyl-17alpha-hydroxyprogesterone acetate medroxyprogesterone acetate 17-Acetoxy-6alpha-methylprogesterone 17alpha-Hydroxy-6alpha-methylprogesterone acetate 6-alpha-Methyl-17-alpha-acetoxyprogesterone 6alpha-Methyl-17-acetoxy progesterone Metigestrona BDBM50067678 (6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-dione 6alpha-Methyl-4-pregnene-3,20-dion-17alpha-ol acetate
- ChEMBL_1570252 (CHEMBL3795163) Inhibition of human CYP17 expressed in Escherichia coli co-transfected with rat NADPH-P450 reductase using progesterone as substrate for 30 mins measured as formation of 17alpha-hydroxyprogesterone and 16alpha-hydroxyprogesterone in presence of NADPH
- ChEMBL_50394 (CHEMBL662850) Ability to inhibit the C17,20-lyase enzyme by 50% using 17-alpha-hydroxyprogesterone as substrate.
- ChEMBL_1275240 (CHEMBL3091212) Inhibition of Wistar rat testicular C17,20-lyase using [3H]17-hydroxyprogesterone as substrate preincubated for 20 mins
- ChEMBL_1614838 (CHEMBL3856907) Inhibition of Wistar rat testicular C17,20-lyase assessed as androst-4-ene-3,17-dione formation using [3H]17-hydroxyprogesterone as substrate in presence of NADPH
- CYP17 Assay The 17 alpha-hydroxylase activity of CYP 17 was determined by measuring the conversion of progesterone into 17 alpha-hydroxyprogesterone and the byproduct 16 alpha-hydroxyprogesterone. Products were analyzed by HPLC. Peak areas were determined by integration of the resulting chromatograms. Substrate conversion was determined by product versus substrate peak areas. The inhibitory potencies were calculated using the diminished substrate conversion caused by the inhibitors.
- CYP17 Inhibition Assay The 17 alpha-hydroxylase activity of CYP 17 was determined by measuring the conversion of progesterone into 17 alpha-hydroxyprogesterone and the byproduct 16 alpha-hydroxyprogesterone. Products were analyzed by HPLC. Peak areas were determined by integration of the resulting chromatograms. Substrate conversion was determined by product versus substrate peak areas. The inhibitory potencies were calculated using the diminished substrate conversion caused by the inhibitors.
- ChEMBL_790050 (CHEMBL1926382) Inhibition of 17,20-lyase activity of Sprague-Dawley rat testicular microsomal CYP17A1 using [1,2-3H]-17a-hydroxyprogesterone as substrate after 15 mins by TLC analysis
- ChEMBL_1675507 (CHEMBL4025650) Inhibition of CYP17 in Sprague-Dawley rat testes microsomes using 17-alpha hydroxyprogesterone as substrate and NADPH as cofactor pretreated for 5 mins followed by substrate and cofactor addition after 60 mins by LC/Ms analysis
- ChEMBL_684688 (CHEMBL1285655) Inhibition of human AKR1C1-mediated progesterone metabolism expressed in bovine aortic endothelial cells assessed as formation of 20alpha-hydroxyprogesterone pretreated for 2 hrs before progesterone challenge measured after 6 hrs by LC/MS analysis
- ChEMBL_1675508 (CHEMBL4025651) Inhibition of recombinant human CYP17 expressed in human A549 cell membranes using 17-alpha hydroxyprogesterone as substrate and NADPH as cofactor pretreated for 5 mins followed by substrate and cofactor addition after 60 mins by LC/MS analysis
- ChEMBL_1625605 (CHEMBL3868074) Inhibition of recombinant CYP21 (unknown origin) overexpressed in human AD293 cells assessed as reduction in 11-deoxycortisol formation preincubated for 60 mins followed by addition of 17-alpha-hydroxyprogesterone as substrate measured after 45 mins by LC-MS/MS analysis
- Enzyme Activity Assay Complementary assays were utilized for the quantitative comparison of compound activity data for CYP17A1 and CYP21A2. Progesterone is a substrate for both CYP17A1 and CYP21A2, and was the substrate studied for enzyme activity, IC50 determinations, and selectivity comparison. Two methods for detecting enzymatic activity were utilized. Analytical High Pressure Liquid Chromatography (HPLC) for biochemical assays was performed on a Prominence HPLC system (Shimadzu Scientific Instruments, Inc., Columbia, Md.) equipped with a C18 reverse phase 100 mm Luna Column (Phenomenex, Torrance, Calif.). The mobile phase consisted of 40% acetonitrile, 59% water, and 1% acetic acid with a 1 mL/min flow rate at 40� C. An injection volume of 32 μL (CYP17A1) or 45 μL (CYP21A2) was used. The presence of the CYP17A1 product 17α-hydroxyprogesterone was detected with an absorption wavelength of 248 nm as reported by Devore, N. M.; Scott, E. E. Structure and function of human cytochromes P450 enzymes: Xenobiotic metabolism by CYP2A and steroid biosynthesis by CYP17A1. University of Kansas, Lawrence, K S, 2011. The presence of the CYP21A2 product 21-hydroxyprogesterone was detected with an absorption wavelength of 248 nm.
- Discontinuous Radiometric Assay Compounds may be evaluated as selective reversible inhibitors of AKR1C3 by screening them against homogeneous recombinant AKR1C1-AKR1C4 expressed in E. coli. In each case, a discontinuous radiometric assay may be used to monitor the inhibition of progesterone reduction (20-ketosteroid reduction) catalyzed by AKR1C1, the inhibition of Δ4-AD reduction (17-ketosteroid reduction) catalyzed by AKR1C3, and the inhibition of 5α-DHT reduction (3-ketosteroid reduction) catalyzed by AKR1C2 and AKR1C4 (by measuring the formation of 20α-hydroxyprogesterone, testosterone or 3α-androstanediol by radiochromatography). Secondary screens of the compounds of interest include: (a) a full-screen against all nine human recombinant AKR enzymes to ensure there are no-intended off-target effects (in this context AKR1B10 (retinal reductase; SEQ ID NO:5) has been shown to be potently inhibited by N-phenylanthranilates) (Endo et al., 2010, Biol. Pharm. Bull. 33:886-90); (b) a screen against COX-1 and COX-2 to reaffirm that compounds do not act as NSAIDs; and (c) an expanded screen against other nuclear receptors (especially other steroid hormone receptors).
 MLS000028438 cid_169870 BDBM70293 SMR000058336 17-alpha-hydroxy-progesterone caproate HYDROXYPROGESTERONE CAPROATE
MLS000028438 cid_169870 BDBM70293 SMR000058336 17-alpha-hydroxy-progesterone caproate HYDROXYPROGESTERONE CAPROATE Rutin caproate BDBM237188
Rutin caproate BDBM237188 BDBM237186 Rutin caproate Rutin capronate
BDBM237186 Rutin caproate Rutin capronate BDBM50423511 HYDROXYPROGESTERONE Delalutin
BDBM50423511 HYDROXYPROGESTERONE Delalutin BDBM50442531 11BETA-HYDROXYPROGESTERONE
BDBM50442531 11BETA-HYDROXYPROGESTERONE n-caproate n-hexylate BDBM50269982 CH3-[CH2]4-COO(-) pentanecarboxylate 1-hexanoate capronate caproate nPnCO2 anion pentylformate hexanoic acid hexoate 1-pentacarboxylate n-hexoate hexylate 1-pentanecarboxylate butylacetate n-Hexanoate hexanoate
n-caproate n-hexylate BDBM50269982 CH3-[CH2]4-COO(-) pentanecarboxylate 1-hexanoate capronate caproate nPnCO2 anion pentylformate hexanoic acid hexoate 1-pentacarboxylate n-hexoate hexylate 1-pentanecarboxylate butylacetate n-Hexanoate hexanoate Methylacetoxyprogesterone US9271961, MPA MEDROXYPROGESTERONE 6-alpha-Methyl-17-alpha-hydroxyprogesterone acetate Medroxyprogesterone 17-acetate (6alpha)-6-methyl-3,20-dioxopregn-4-en-17-yl acetate Medroxyacetate progesterone 6alpha-Methyl-17alpha-hydroxyprogesterone acetate medroxyprogesterone acetate 17-Acetoxy-6alpha-methylprogesterone 17alpha-Hydroxy-6alpha-methylprogesterone acetate 6-alpha-Methyl-17-alpha-acetoxyprogesterone 6alpha-Methyl-17-acetoxy progesterone Metigestrona BDBM50067678 (6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-dione 6alpha-Methyl-4-pregnene-3,20-dion-17alpha-ol acetate
Methylacetoxyprogesterone US9271961, MPA MEDROXYPROGESTERONE 6-alpha-Methyl-17-alpha-hydroxyprogesterone acetate Medroxyprogesterone 17-acetate (6alpha)-6-methyl-3,20-dioxopregn-4-en-17-yl acetate Medroxyacetate progesterone 6alpha-Methyl-17alpha-hydroxyprogesterone acetate medroxyprogesterone acetate 17-Acetoxy-6alpha-methylprogesterone 17alpha-Hydroxy-6alpha-methylprogesterone acetate 6-alpha-Methyl-17-alpha-acetoxyprogesterone 6alpha-Methyl-17-acetoxy progesterone Metigestrona BDBM50067678 (6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-dione 6alpha-Methyl-4-pregnene-3,20-dion-17alpha-ol acetate